Viêm quanh implant: so sánh phương pháp
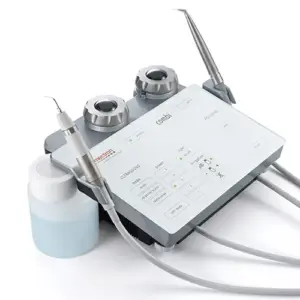
The Mectron Prophylaxis Combi Touch in the Non-Surgical Management of Peri-Implantitis: A Clinical and Technical Review
The Pathophysiology and Diagnosis of Peri-Implantitis
From Peri-Implant Mucositis to Peri-Implantitis: An Inflammatory Continuum
The long-term success of dental implants is threatened by inflammatory conditions collectively known as peri-implant diseases. These conditions exist on a spectrum, beginning with a reversible inflammatory state and progressing to irreversible tissue destruction. Understanding this continuum is fundamental to timely diagnosis and appropriate therapeutic intervention.1 The initial stage, peri-implant mucositis, is defined as a reversible inflammatory lesion confined to the soft tissues surrounding a dental implant.2 Clinically, it is characterized by signs of inflammation such as redness and swelling, and most notably, bleeding on gentle probing (BOP).3 This condition is analogous to gingivitis around natural teeth and is considered the direct precursor to the more destructive peri-implantitis.1 Crucially, peri-implant mucositis does not involve progressive bone loss beyond the initial physiological bone remodeling that occurs after implant placement.5 Because it is reversible, early detection and treatment of mucositis are paramount in preventing the onset of peri-implantitis.1 If left untreated, peri-implant mucositis can advance to peri-implantitis, a more severe pathological state. Peri-implantitis is characterized by inflammation of the peri-implant connective tissues coupled with the progressive and irreversible loss of supporting alveolar bone.6 This loss of osseointegration compromises the stability of the implant and, without intervention, can ultimately lead to implant failure and loss.2 The biological differences between the peri-implant and periodontal tissues—specifically the absence of a periodontal ligament and a less robust vascular supply around an implant—can result in an inflammatory lesion that progresses more rapidly and destructively than in periodontitis. This underscores the critical need for implant-specific diagnostic and therapeutic protocols.
Etiology and Key Risk Factors in Disease Progression
The primary etiological factor for both peri-implant mucositis and peri-implantitis is the accumulation of a pathogenic bacterial biofilm on the implant surface.1 This biofilm triggers a host inflammatory response that, in susceptible individuals, leads to tissue destruction.1 The micro-roughened surfaces of modern dental implants, while beneficial for osseointegration, are particularly susceptible to biofilm accumulation and are challenging to decontaminate once colonized.10 While biofilm is the initiator, a confluence of systemic and local factors significantly increases the risk of disease development and progression. A comprehensive risk assessment is therefore essential for patient selection and long-term management. Key contributing factors include:
- Poor Oral Hygiene: Inadequate plaque control is a primary driver of biofilm accumulation and is strongly associated with the incidence of peri-implant diseases.2
- History of Periodontitis: Patients with a history of periodontal disease are at a significantly higher risk of developing peri-implantitis, suggesting a heightened host susceptibility to pathogenic biofilms.6
- Smoking and Tobacco Use: Smoking is a major risk factor that impairs host immune response and wound healing, exacerbating the destructive processes of peri-implantitis.2
- Systemic Conditions: Uncontrolled systemic diseases, such as diabetes mellitus, can alter the inflammatory response and increase susceptibility to infection. Other conditions like osteoporosis may also play a role.6
- Prosthetic Factors: Ill-fitting restorations, open margins, or the presence of residual submucosal cement can create niches for bacterial accumulation and impede effective oral hygiene, thereby increasing disease risk.4
- Biomechanical Overload: Excessive occlusal forces, often associated with bruxism, can cause micro-damage to the implant and surrounding bone, potentially creating pathways for bacterial invasion and accelerating bone loss.2
Standardized Clinical and Radiographic Diagnostic Criteria
Accurate and early diagnosis is critical for successful management. A standardized set of clinical and radiographic criteria has been established to differentiate between peri-implant health, mucositis, and peri-implantitis. Key clinical signs of peri-implantitis include bleeding on probing (BOP) and/or suppuration (the discharge of pus), an increase in probing pocket depth (PD) compared to baseline, and visible inflammation of the peri-implant tissues, such as redness, swelling (edema), and tenderness.2 In advanced cases, the implant may exhibit detectable mobility, which is a sign of severe bone loss and imminent failure.2 The consensus diagnostic criteria depend on the availability of baseline data, which ideally should be recorded after the initial healing and prosthetic loading phase. This distinction highlights a significant challenge in clinical practice, as many patients present for care without historical records. Consequently, clinicians must often rely on absolute thresholds for diagnosis. This reliance on static measurements, rather than tracking change over time, can complicate case assessment but underscores the need for a versatile therapeutic approach capable of managing a range of clinical presentations.
- Diagnosis with Baseline Data: Peri-implantitis is diagnosed in the presence of BOP and/or suppuration, a documented increase in probing depth from baseline, and radiographic evidence of bone loss beyond the initial crestal bone remodeling.5
- Diagnosis without Baseline Data: In the absence of previous records, a diagnosis of peri-implantitis can be made based on the combination of BOP and/or suppuration, a probing pocket depth of $6$ mm or greater, and radiographic evidence of bone levels located $3$ mm or more apical to the most coronal portion of the intraosseous part of the implant.11
Table 1: Diagnostic Criteria for Peri-Implant Health, Mucositis, and Peri-Implantitis Condition Clinical Signs (BOP/Suppuration) Clinical Signs (Probing Depth) Radiographic Bone Loss Peri-Implant Health No bleeding or suppuration No increase from baseline No loss beyond initial remodeling Peri-Implant Mucositis Bleeding and/or suppuration present May or may not have increased PD No loss beyond initial remodeling Peri-Implantitis Bleeding and/or suppuration present Increased PD from baseline Progressive loss beyond initial remodeling*
*In the absence of baseline data, a probing depth of $\ge6$ mm is considered indicative. 11
**In the absence of baseline data, bone levels $\ge3$ mm apical to the implant-abutment junction are considered indicative. 11
The Rationale and Objectives of Non-Surgical Peri-Implantitis Therapy
Therapeutic Goals: Biofilm Disruption and Implant Surface Decontamination
The fundamental goal of non-surgical peri-implantitis therapy is to control the underlying infection and halt the progression of the disease.8 This is achieved through the meticulous disruption and removal of the pathogenic biofilm and calcified deposits (calculus) from the implant surface, abutment, and surrounding soft tissues.2 This process, known as surface decontamination, aims to reduce the bacterial load to a level that is compatible with tissue health, thereby arresting the inflammatory process and preventing further bone loss.13 The ultimate objective is to preserve the function and longevity of the osseointegrated implant.8
An Overview of Non-Surgical Modalities and Their Limitations
The cornerstone of non-surgical treatment is mechanical debridement, which involves the physical removal of plaque and calculus.2 Several modalities exist, each with distinct advantages and significant limitations, particularly concerning the delicate nature of the titanium implant surface.
- Hand Instruments: Curettes made from materials like titanium or carbon fiber have been traditionally used.1 While effective, they are technically demanding to use in the complex geometry of an implant and can inadvertently scratch or alter the implant surface, creating new niches for plaque accumulation.16
- Ultrasonic Scalers: These power-driven instruments are efficient for removing calculus. However, conventional stainless-steel ultrasonic tips are significantly harder than titanium and can cause severe surface damage, including scratches and gouging.16 This iatrogenic damage can compromise the biocompatibility of the implant and accelerate disease recurrence.
- Air Polishing: This modality uses a high-velocity stream of air, water, and an abrasive powder. Traditional powders, such as sodium bicarbonate, are highly effective for stain removal on enamel but are too abrasive for subgingival use and can damage both root surfaces and titanium implant surfaces.18
This presents a central therapeutic paradox: the very act of cleaning can cause damage that perpetuates the disease cycle. The instruments designed to remove biofilm can increase surface roughness, which in turn enhances future biofilm adhesion and retention.19 The development of implant-safe materials, such as PEEK for ultrasonic tips and low-abrasion powders like glycine, represents a fundamental solution to this paradox, enabling effective decontamination without causing iatrogenic harm. Adjunctive therapies, such as the local application of antimicrobial rinses or gels containing chlorhexidine, are often employed to supplement mechanical debridement.2 However, their clinical benefit remains a subject of debate, with some evidence suggesting they provide no significant additional effect over thorough mechanical cleaning alone.20
The Role of Non-Surgical Therapy in the Overall Treatment Sequence
There is a broad consensus within clinical guidelines that non-surgical therapy should always be the first-line treatment for peri-implantitis.1 This initial phase is a mandatory prerequisite before any surgical intervention is considered.15 The rationale for this sequential approach is twofold. First, it serves a direct therapeutic purpose. In cases of peri-implant mucositis and early peri-implantitis, a thorough non-surgical intervention may be sufficient to resolve the inflammation and arrest disease progression, obviating the need for more invasive procedures.1 Second, and equally important, this initial phase functions as a critical diagnostic and prognostic tool. The patient's response to non-surgical therapy provides invaluable information. It allows the clinician to assess the patient's ability and willingness to perform effective home care, which is a non-negotiable factor for long-term success.4 Furthermore, the tissue's healing response helps to determine the true nature of the underlying defect. If the established therapeutic endpoints—such as a residual probing depth of $\le5$ mm and the absence of bleeding on probing—are not achieved after a well-executed non-surgical protocol, it indicates that the defect morphology or bacterial load is too complex for a closed approach.12 This "failure" of non-surgical therapy is not a failure of the clinician but rather a confirmation that surgical access is required. Therefore, non-surgical therapy acts as a "diagnostic filter," effectively treating amenable cases while accurately identifying refractory cases that require surgery.
The Mectron Prophylaxis Combi Touch: A Comprehensive Technical Analysis
The Mectron Prophylaxis Combi Touch is an advanced clinical unit engineered to address the specific challenges of modern periodontal and peri-implant therapy. Its design integrates two essential modalities—piezoelectric ultrasound and air polishing—into a single, synergistic system, facilitating a comprehensive and minimally invasive treatment protocol.
The Dual-Modality System: Integrated Piezoelectric Ultrasound and Air Polishing
The Combi Touch consolidates the functions of a high-precision ultrasonic scaler and a versatile air-polishing device into one unit.21 This integration allows the clinician to perform a complete prophylaxis treatment, seamlessly transitioning from the removal of hard calculus deposits to the gentle management of soft biofilm on natural teeth, restorations, and dental implants.23 The device is controlled via an ergonomic, smooth-surface touch panel, which not only provides an intuitive user interface but also allows for rapid and effective cleaning and disinfection between patients, adhering to modern infection control standards.21
Ultrasonic System Features: Precision and Patient Comfort
The ultrasonic component of the Combi Touch is designed for efficacy, safety, and patient comfort, incorporating several key technologies.
- Piezoelectric Principle: The unit operates on a piezoelectric principle, which generates controlled, linear (back-and-forth) oscillations of the instrument tip.24 This is in contrast to magnetostrictive systems, which produce a more aggressive elliptical motion. The linear movement is gentler on tooth and implant surfaces and is generally perceived as more comfortable by the patient, as it avoids any unpleasant hammering sensation.24
- SOFT MODE: A standout feature is the innovative SOFT MODE, which can be activated to reduce the amplitude of the ultrasonic oscillations.21 This allows the insert to move with a gentler yet still efficient action, making it ideal for treating sensitive patients and, critically, for debriding delicate implant surfaces where minimizing physical impact is paramount.25
- Feedback System: The device incorporates an intelligent feedback system that continuously monitors the resistance encountered by the insert tip and automatically adjusts the power output in real-time.23 This ensures that the optimal power level is delivered for the specific clinical task, preventing the application of excessive force while maintaining consistent performance.25
- LED Handpiece: The slim, lightweight (55 g) ultrasonic handpiece is equipped with a circular, 360° rotatable LED light.21 This design provides brilliant, focused illumination directly at the working tip, regardless of the insert's shape or angle, significantly enhancing visibility in all areas of the oral cavity.25
Air-Polishing System Features: Safety and Subgingival Access
The air-polishing system is engineered for versatility, with a strong emphasis on safety during subgingival application. The true innovation lies not merely in combining two technologies, but in the synergistic integration of features that support a clinical philosophy of minimally invasive, patient-centric peri-implant care. The SOFT MODE on the ultrasonic side, for example, is the conceptual partner to the PERIO Function on the air-polishing side; both are designed to reduce aggressiveness for sensitive applications. This integrated design facilitates a protocol-driven approach where the technology is tailored to the goal of maximum efficacy with minimum iatrogenic damage.
- Supra- and Subgingival Modes: The clinician can instantly switch between a standard supragingival "prophy" mode and a specialized subgingival "perio" mode at the touch of a button.21
- PERIO Function: This is a critical safety feature. When activated, the PERIO function significantly reduces the air pressure of the powder jet.25 This controlled, low-pressure delivery is essential for the safe and gentle removal of biofilm from within periodontal and peri-implant pockets, minimizing the risk of soft tissue trauma or iatrogenic facial emphysema.26
- Dual Powder Chambers: The unit is equipped with two easily removable powder chambers, facilitating the use of different powders within the same procedure.24 A common workflow involves using sodium bicarbonate in the "prophy" chamber for heavy stain removal on enamel and a low-abrasive glycine powder in the "perio" chamber for subgingival and implant applications.28 An exclusive "refill" function depressurizes the chambers, allowing them to be removed for cleaning or refilling without powering down the entire unit.21
- Independent Irrigation System: A 500 ml bottle system provides the irrigation fluid, allowing for the use of water or specific medicinal solutions, such as chlorhexidine for antimicrobial therapy or sodium hypochlorite for endodontic procedures.21 The water can also be heated to a comfortable physiological temperature, enhancing patient comfort.25
- Clogging Protection: To ensure consistent and reliable performance, a constant stream of light air is maintained through the tubing whenever the unit is powered on. This prevents powder from clumping and causing blockages.21
Table 2: Key Features of the Mectron Combi Touch for Peri-Implant Therapy
Feature Clinical Relevance for Peri-Implantitis PEEK-Coated Ultrasonic Tips Biocompatible polymer is softer than titanium, allowing for calculus removal without scratching or damaging the implant surface.29 SOFT MODE (Ultrasonic) Reduces the amplitude of ultrasonic oscillations for a gentler debridement, further minimizing the risk of surface alteration on implants and enhancing comfort for sensitive patients.21 Glycine Powder Compatibility Low-abrasion powder effectively removes biofilm from implant surfaces, restorations, and subgingival areas without causing significant surface damage.25 PERIO Function (Air Polishing) Reduces air pressure for safe subgingival biofilm removal, minimizing the risk of soft tissue trauma and preventing complications like subcutaneous emphysema.26 Subgingival Perio Tip A flexible, sterile, disposable tip designed for safe access and effective decontamination of peri-implant pockets deeper than 5 mm.24 Independent Bottle System Allows for the use of therapeutic irrigants, such as chlorhexidine, to provide adjunctive antimicrobial action during the decontamination procedure.21
Specialized Instrumentation for Safe and Effective Implant Decontamination
The efficacy and safety of any non-surgical peri-implant therapy are ultimately determined by the instruments that make direct contact with the implant surface. The Mectron Combi Touch system utilizes a suite of specialized, purpose-built tips, nozzles, and powders designed to achieve thorough decontamination while preserving the integrity of the delicate titanium surface.
Ultrasonic Instrumentation: The Role of PEEK-Coated Tips (IC1)
To overcome the significant risk of surface damage posed by traditional metal ultrasonic scalers, Mectron has developed implant-specific cleaning tips made from PEEK (polyetheretherketone).29 PEEK is an advanced, biocompatible thermoplastic polymer known for its high mechanical strength and wear resistance, yet it is significantly softer than titanium alloy.30 This critical material property allows for the effective removal of calculus and plaque from implant surfaces and restorations without causing the scratches, pitting, or contamination associated with harder materials.32 Mectron's "Implant cleaning set S" features a modular design, consisting of a reusable, medical-grade stainless steel tip holder (designated ICS or ICP) and replaceable, single-use or autoclavable PEEK tips (designated IC1).29 The IC1 tip is conically shaped and attached to a 102° angled holder, providing excellent access for cleaning around implant abutments.29 This design is also cost-effective, as only the relatively inexpensive PEEK tip needs to be replaced when it wears, rather than the entire metal insert.30
Air-Polishing Modality: The Clinical Advantages of Low-Abrasive Glycine Powder
For the air-polishing modality, the choice of powder is paramount. The Combi Touch system is designed to be used with low-abrasive powders for subgingival and peri-implant applications, specifically glycine.24 Glycine, a naturally occurring amino acid, has physical properties that make it ideal for this purpose. Its particles are significantly smaller (average size $<25$ μm) and softer than those of traditional sodium bicarbonate.28 This low abrasiveness allows glycine powder to be used safely and effectively for removing biofilm from sensitive surfaces, including exposed root dentin, restorative materials, and, most importantly, titanium implant surfaces, without causing clinically significant abrasion or alteration.25 As biofilm is the primary etiological factor in peri-implantitis, the ability to thoroughly remove it without damaging the underlying surface is the central goal of non-surgical therapy.24 Furthermore, glycine is water-soluble and has a mildly sweet taste, which improves patient acceptance and comfort compared to the salty taste of sodium bicarbonate.23 The Combi Touch unit is supplied with "mectron prophylaxis powder sensitive+," a glycine-based formulation specifically intended for these applications.24
Nozzle and Tip Selection for Varying Pocket Depths and Anatomies
Effective and safe subgingival debridement requires instrumentation that can be adapted to the specific anatomy of the peri-implant pocket. A rigid, wide-bore nozzle suitable for supragingival cleaning cannot safely access a deep, narrow peri-implant defect. This recognition of depth-specific challenges represents a significant evolution in non-surgical therapy. The Combi Touch system provides a protocolized, safety-oriented solution with a range of purpose-built nozzles and tips.
- For Pockets $\le5$ mm: For supra- and subgingival cleaning in shallow to moderate pockets (up to 5 mm deep), the system utilizes rigid 90° or 120° angled spray nozzles.24 The manufacturer's clinical protocol specifically recommends the 120° nozzle for treating peri-implantitis in this depth range.24
- For Pockets $>5$ mm: For safe and effective access into deeper pockets, a specialized PERIO spray head is required.24 This head is designed to accept a disposable, sterile, and flexible Subgingival Perio Tip.25 This soft, narrow tip can be gently inserted into the pocket, allowing the low-pressure glycine spray to reach the base of the defect for thorough decontamination without traumatizing the surrounding soft tissues.24 This depth-specific approach enhances both the safety and predictability of treating more advanced non-surgical cases.
A Step-by-Step Clinical Protocol for Non-Surgical Treatment of Peri-Implantitis
The therapeutic potential of the Mectron Combi Touch is realized not through the device alone, but through the disciplined execution of a multi-step, multi-modality clinical protocol. This protocol synthesizes the device's technical capabilities with evidence-based principles of implant surface decontamination. The clinician's diagnostic accuracy and procedural adherence are paramount to achieving predictable outcomes.
Patient Preparation and Initial Debridement with PEEK-Tipped Ultrasonics
1. Pre-procedural Preparation: The treatment should begin with the patient performing a pre-procedural rinse with an antimicrobial agent, such as a 0.12% chlorhexidine gluconate solution, to reduce the overall microbial load in the oral cavity.11 2. Anesthesia: Local anesthesia should be administered as indicated to ensure patient comfort, particularly if deep pockets or inflamed tissues are present.11 3. Calculus Removal: The first active treatment step is the removal of any hard calculus deposits from the exposed surfaces of the implant and abutment.24 This task requires the mechanical energy of an ultrasonic scaler. 4. Instrumentation: The Mectron ultrasonic handpiece should be fitted with an IC1 PEEK-coated tip.29 The clinician should activate the SOFT MODE on the Combi Touch unit to deliver gentle, low-amplitude oscillations.21 The instrument should be used with light pressure to methodically debride all accessible surfaces, ensuring complete calculus removal without scratching or altering the underlying titanium.
Subgingival Biofilm Detoxification with Glycine Powder Air Polishing
1. Device Setup: The "perio" powder chamber should be confirmed to be filled with Mectron's glycine-based powder ("sensitive+").24 2. Mode Selection: The clinician must select the "perio" function on the device's touch panel. This is a critical safety step that automatically reduces the system's air pressure to a level appropriate for subgingival application.26 3. Clinical Rationale: After the removal of hard deposits, this step targets the primary etiological factor: the residual pathogenic biofilm covering the implant surface within the peri-implant pocket.24 Clinical evidence demonstrates that this dual-modality approach, targeting both calculus and biofilm, is significantly more effective than ultrasonic debridement alone.37
Technique Modifications for Pocket Depths ≤5 mm versus >5 mm
The application technique for glycine powder air polishing must be adapted based on the diagnosed probing depth to ensure both safety and efficacy.
- For Pockets $\le5$ mm:
- Instrumentation: Attach the 120° angled spray nozzle to the air-polishing handpiece.24
- Technique: The nozzle tip is directed towards the implant surface and gently advanced into the sulcus or pocket. The spray is activated for a limited duration (e.g., 5 seconds per aspect) to decontaminate the implant surface and disrupt the biofilm within the pocket.11
- For Pockets $>5$ mm:
- Instrumentation: Attach the dedicated PERIO spray nozzle to the handpiece, followed by a new, sterile, disposable Subgingival Perio Tip.24
- Technique: This step requires a specific and careful technique. The flexible Perio Tip is gently inserted into the deep pocket. Crucially, the handpiece and tip must be held parallel to the long axis of the implant during activation.24 This orientation ensures that the low-pressure spray is directed apically along the implant surface to decontaminate the base of the defect and flush out biofilm and granulation tissue, minimizing the risk of tissue trauma or emphysema.24
Post-Treatment Care and Maintenance Recommendations
1. Post-procedural Irrigation: Following decontamination, the treated pockets should be gently irrigated with a sterile saline solution or a prescribed antimicrobial agent to flush out any remaining debris.11 2. Oral Hygiene Instruction (OHI): The success of the treatment is critically dependent on the patient's long-term maintenance. Individualized OHI must be provided and reinforced, focusing on effective techniques for cleaning around the implant prosthesis using tools such as interdental brushes or floss.2 3. Supportive Peri-Implant Care (SPIC): The patient must be enrolled in a regular professional maintenance program. A typical recall interval is every 3 months, especially in the first year post-treatment.8 These appointments are essential for monitoring tissue health, reinforcing OHI, and performing professional debridement to prevent disease recurrence.
Clinical Evidence and Comparative Efficacy Analysis
The clinical utility of the technologies integrated into the Mectron Combi Touch—specifically PEEK-tipped piezoelectric ultrasonics and glycine powder air polishing (GPAP)—is supported by a growing body of scientific literature, including randomized controlled trials (RCTs) and systematic reviews.
Review of Randomized Controlled Trials (RCTs): GPAP vs. Conventional Debridement
Comparative studies have evaluated the efficacy of GPAP against other non-surgical debridement methods for both peri-implant mucositis and peri-implantitis. For peri-implant mucositis, a 12-month RCT found that treatment with either GPAP or a conventional ultrasonic (US) device was equally effective. Both groups showed statistically significant reductions in mean plaque scores, bleeding on probing, and the number of pockets $\ge4$ mm compared to baseline, indicating that GPAP is a valid and effective modality for managing this early-stage disease.38 For the treatment of peri-implantitis, a 2-month RCT yielded more nuanced results. While both GPAP and an ultrasonic device led to significant improvements in probing depth (PD), BOP, and plaque index (PI), the GPAP group demonstrated a more pronounced reduction in inflammation. The mean BOP score in the GPAP group decreased from 94.4% to 57.8%, whereas the US group saw a reduction from 91.1% to 72.2%.3 This suggests a potential superiority of GPAP in resolving soft tissue inflammation. Regarding patient experience, an RCT directly comparing the Mectron Combi Touch with the EMS Prophylaxis Master during guided biofilm therapy sessions found comparable overall levels of patient and operator comfort. Notably, the Mectron Combi Touch was reported to have higher comfort specifically related to the airflow component of the treatment.40 Both systems were equally effective at reducing periodontal parameters.
The Synergy of PEEK-Tipped Ultrasonics and GPAP in Supportive Care Protocols
Perhaps the most compelling evidence for a dual-modality approach comes from a landmark 12-month RCT that investigated a supportive peri-implant care (SPIC) protocol following surgical treatment for peri-implantitis.37 This study isolated the effect of GPAP when added to a baseline of safe ultrasonic debridement.
- The Test Group received debridement with a piezoelectric ultrasonic scaler using a PEEK tip, followed by air polishing with glycine powder (GPAP).
- The Control Group received the same PEEK-tipped ultrasonic debridement, but this was followed by polishing with a traditional rubber cup and paste.
The results were striking. After 12 months, the test group (ultrasonics + GPAP) demonstrated a statistically significant mean probing depth reduction of $-0.84$ mm. In stark contrast, the control group (ultrasonics alone) experienced a mean probing depth increase of $+0.18$ mm. The difference between the groups was highly significant ($p <.001$).37 This finding is profound. It demonstrates that safe removal of calculus with PEEK-tipped ultrasonics, while necessary, is insufficient on its own to maintain peri-implant health or prevent disease recurrence in a supportive care context. The deterioration of the control group suggests that the residual biofilm left after scaling is enough to drive disease progression. The significant improvement in the test group confirms that the additional, effective removal of this biofilm with GPAP is the critical factor in halting and even reversing the disease process. This provides a powerful, evidence-based rationale for adopting the integrated, dual-modality protocol facilitated by the Combi Touch system.
Meta-Analysis Insights: Positioning Air Abrasion in the Therapeutic Armamentarium
A recent systematic review and meta-analysis provides a broader perspective on the role of air abrasion. The analysis concluded that, overall, air abrasion was just as effective as other mechanical decontamination methods (e.g., curettes, ultrasonic scalers) in reducing probing depth and alveolar bone loss during the initial non-surgical treatment of peri-implantitis.10 However, the subgroup analysis revealed important nuances. Air abrasion was found to be less effective than other methods in reducing probing depth when used for long-term maintenance purposes, and less effective in reducing bone loss when used in surgical approaches.10 This suggests that the primary strength of air abrasion lies in the initial, comprehensive non-surgical debridement phase. It is also important to consider that some in-vitro studies report lower "cleaning efficacy" for air polishing compared to titanium brushes or ultrasonic scalers.14 This likely reflects the inability of air polishing to remove hard calculus, reinforcing its role as a specialized tool for biofilm management that should be used in conjunction with, not as a replacement for, ultrasonic scaling. Table 3: Summary of Key Clinical Trial Outcomes for Glycine Powder Air Polishing (GPAP) and PEEK-Tipped Ultrasonics
Study Reference Condition Treated Test Group Intervention Control Group Intervention Key Outcomes Follow-up Monje et al. 2023 37 Peri-implantitis (Supportive Care) PEEK-tipped US + GPAP PEEK-tipped US + Rubber Cup PD Reduction: -0.84 mm (Test) vs. +0.18 mm (Control) ($p <.001$) 12 months Memè et al. 2023 3 Peri-implantitis Glycine Powder Air Polishing (GPAP) Ultrasonic (US) Device BOP Reduction: -36.6% (GPAP) vs. -18.9% (US) 2 months Sculean et al. 2015 38 Peri-implant Mucositis Glycine Powder Air Polishing (GPAP) Ultrasonic (US) Debridement Both groups showed significant and comparable reductions in PI, BOP, and PD. 12 months Butera et al. 2024 40 Periodontal Maintenance (GBT) Mectron Combi Touch EMS Prophylaxis Master Comparable patient/operator comfort and reduction in periodontal parameters. N/A
Patient Selection and Clinical Risk Management
The successful implementation of non-surgical peri-implantitis therapy using the Mectron Combi Touch requires careful patient selection, a thorough understanding of contraindications, and diligent management of potential complications. While the device incorporates advanced safety features, the ultimate responsibility for safe and effective treatment rests with the trained clinician.
Criteria for Patient Selection in Non-Surgical Peri-Implantitis Therapy
Non-surgical therapy is the indicated first-line treatment for patients diagnosed with peri-implant mucositis and early-to-moderate peri-implantitis.5 The ideal candidate presents with a defect morphology that is accessible to closed instrumentation. However, two essential prerequisites must be met before initiating therapy: 1. Patient Motivation and Oral Hygiene Capability: The patient must demonstrate the ability and commitment to perform adequate self-care.4 Non-surgical therapy is unlikely to succeed in the long term without excellent daily plaque control by the patient. Therefore, treatment should be deferred in unmotivated patients until their oral hygiene practices improve.12 2. Prosthesis Cleansability: The implant-supported prosthesis must be designed to allow the patient and clinician adequate access for cleaning. If the contours of the restoration are over-bulked or margins are poor, the prosthesis should be modified or remade prior to initiating definitive therapy.1
Systemic and Local Contraindications for Ultrasonic and Air-Polishing Procedures
Certain systemic and local conditions may contraindicate or require modification of the treatment protocol.
- General Contraindications for Air Polishing:
- Respiratory Conditions: Patients with severe or unstable respiratory diseases, such as chronic obstructive pulmonary disease (COPD) or uncontrolled asthma, may be poor candidates due to the aerosol generated.19
- Infectious Diseases: Caution is required for patients with infectious diseases that can be transmitted via aerosols.19
- Allergies: Patients with known allergies to the powder components (e.g., glycine) should not be treated with that specific powder.42
- Powder-Specific Contraindications:
- The use of sodium bicarbonate powder is contraindicated for patients on sodium-restricted diets (due to conditions like hypertension or renal disease) and should not be used on exposed root surfaces or most composite and glass ionomer restorations.18 This is less of a concern for the peri-implantitis protocol, which mandates the use of glycine.
- Local Contraindications:
- Treatment should be approached with caution in areas with minimal or no attached keratinized mucosa or in the presence of extremely inflamed, friable tissue. These situations may increase the risk of procedural complications like emphysema.42
Identification and Management of Potential Complications
While modern devices are engineered for safety, the clinician must be aware of potential complications and the strategies to prevent them.
- Subcutaneous Emphysema: This is a rare but potentially serious complication where air from the air-polishing device is forced into the subcutaneous or fascial spaces of the head and neck, causing rapid swelling and discomfort.17 The risk is significantly mitigated by the Combi Touch's built-in safety features, but prevention is operator-dependent. The clinician must:
- Always use the low-pressure PERIO function for any subgingival application.26
- Use the correct instrumentation (e.g., the Subgingival Perio Tip for deep pockets).25
- Maintain proper nozzle angulation, keeping the tip parallel to the implant axis and avoiding directing the jet directly into the base of the pocket.17
- Implant Surface Alteration: The risk of scratching or damaging the implant surface is a primary concern. This is prevented by strict adherence to the use of implant-safe instrumentation: only PEEK-coated tips for ultrasonic scaling and only low-abrasion glycine powder for air polishing on any implant or restorative surface.16
- Aerosol Production: Both ultrasonic scaling and air polishing generate significant aerosols, posing a risk of disease transmission.19 Management requires adherence to standard infection control protocols, including the mandatory use of high-volume evacuation (HVE), pre-procedural rinses, and appropriate personal protective equipment (PPE) for both the patient and the clinical team.19
Table 4: Contraindications and Risk Mitigation for Non-Surgical Peri-Implant Debridement
Risk/Contraindication Associated Modality Mitigation Strategy / Recommended Action Severe Respiratory Disease Air Polishing Consult physician; consider alternative debridement methods (e.g., ultrasonic only). Sodium-Restricted Diet Air Polishing (Prophy Mode) Use only glycine or other sodium-free powders; strictly avoid sodium bicarbonate. Subcutaneous Emphysema Air Polishing (Perio Mode) Use PERIO function (low pressure), select depth-appropriate nozzle/tip, maintain correct parallel angulation to the implant axis.17 Implant Surface Damage Ultrasonic Scaling, Air Polishing Use only PEEK-coated ultrasonic tips. Use only low-abrasion glycine powder. Never use metal tips or sodium bicarbonate on implant surfaces.16 Aerosol Contamination Ultrasonic Scaling, Air Polishing Mandatory use of high-volume evacuation (HVE), pre-procedural rinse, and full PPE for patient and operator.19
Clinical Recommendations and Future Perspectives
The comprehensive analysis of the Mectron Prophylaxis Combi Touch system, its specialized instrumentation, and the supporting clinical evidence allows for the formulation of clear, evidence-based recommendations for its integration into modern dental practice for the management of peri-implant diseases.
Evidence-Based Recommendations for Integrating the Combi Touch into Clinical Practice
1. Adopt a Dual-Modality Protocol: It is strongly recommended that clinicians adopt a sequential, dual-modality protocol for the non-surgical treatment of peri-implantitis. This protocol should begin with the removal of hard deposits using PEEK-tipped piezoelectric ultrasonics, immediately followed by the detoxification of the implant surface with glycine powder air polishing to remove the pathogenic biofilm. Evidence suggests this combined approach is superior to either modality used in isolation.37 2. Mandate Implant-Safe Instrumentation: The use of instrumentation specifically designed to be safe for titanium surfaces should be considered mandatory, not optional. This includes the exclusive use of PEEK-coated tips (e.g., Mectron IC1) for any ultrasonic scaling on or near an implant and the use of low-abrasion powders (e.g., glycine) for all air-polishing procedures on implants and restorations. This practice is essential to prevent iatrogenic surface damage that can perpetuate the disease cycle.16 3. Utilize for Supportive Peri-Implant Care (SPIC): The Combi Touch system is highly indicated for use in routine SPIC protocols. The evidence from long-term supportive care trials highlights that effective biofilm removal with glycine powder air polishing is critical for preventing disease recurrence and maintaining peri-implant health following active treatment.37
Summary of Best Practices for Predictable Outcomes
To achieve predictable and successful outcomes in non-surgical peri-implantitis therapy, clinicians should adhere to the following best practices:
- Foundation in Diagnosis: Begin with an accurate diagnosis based on standardized criteria, and perform meticulous charting of baseline parameters (PD, BOP, radiographic bone levels) whenever possible.
- Prioritize Patient Factors: Patient selection must prioritize individuals who are motivated and capable of maintaining a high standard of oral hygiene. Address prosthetic issues that impede cleansability before initiating therapy.
- Adhere to Protocol: Strictly follow the depth-specific instrumentation protocol, using the appropriate nozzles and tips (e.g., 120° nozzle for pockets $\le5$ mm, Subgingival Perio Tip for pockets $>5$ mm) and safety features (PERIO function, SOFT MODE).
- Sequential Treatment Planning: View non-surgical therapy as the indispensable first step in a comprehensive treatment algorithm. This phase must be followed by a formal re-evaluation to assess treatment outcomes and determine the need for subsequent surgical intervention or transition to a supportive care phase.
Future Directions in Non-Surgical Peri-Implant Disease Management
The field of peri-implant disease management continues to evolve. Future research and development will likely focus on several key areas:
- Long-Term Clinical Trials: While short- and mid-term data are promising, there is a need for more multi-center, long-term RCTs to confirm the durability of clinical improvements achieved with advanced non-surgical protocols and to compare them directly against surgical interventions.
- Biomarker-Guided Therapy: The analysis of biomarkers in the peri-implant crevicular fluid, such as inflammatory cytokines or enzymes related to bone metabolism, may one day provide a more objective means of monitoring disease activity, assessing treatment response, and predicting patient risk, allowing for more personalized therapy.9
- Advancements in Materials and Technology: The continued development of novel, even less abrasive powders (such as erythritol) and more ergonomically advanced and intelligent delivery systems will further refine the safety and efficacy of non-surgical approaches, potentially expanding the range of cases that can be successfully managed without surgery.14
Nguồn trích dẫn 1. Non-Surgical Management of Peri-Implant Diseases – ResearchGate, truy cập vào tháng 10 24, 2025, https://www.researchgate.net/profile/Andrea-Roccuzzo/publication/351955988_Non-Surgical_Management_of_Peri-implant_Diseases/links/60b20964a6fdcc1c66ebf699/Non-Surgical-Management-of-Peri-implant-Diseases.pdf 2. What is Peri-implantitis and What to Do About It? – MD Periodontics, truy cập vào tháng 10 24, 2025, https://mdperio.com/blog/what-is-peri-implantitis/ 3. (PDF) Evaluation of a single non-surgical approach in the …, truy cập vào tháng 10 24, 2025, https://www.researchgate.net/publication/388867792_Evaluation_of_a_single_non-surgical_approach_in_the_management_of_peri-implantitis_glycine_powder_air-polishing_versus_ultrasonic_device 4. Peri-implant disease: early diagnosis and non-surgical treatment—a narrative literature review, truy cập vào tháng 10 24, 2025, https://www.monadnockperio.com/wp-content/uploads/2021/02/peri-implant-disease.pdf 5. Diagnosis and non-surgical treatment of peri-implant diseases and maintenance care of patients with dental implants – Consensus report of working group 3 – NIH, truy cập vào tháng 10 24, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC9379037/ 6. Peri-Implantitis Symptoms, Causes, Diagnosis, Treatment, truy cập vào tháng 10 24, 2025, https://www.precisioninperio.com/peri-implantitis-symptoms-causes-diagnosis-treatment/ 7. Peri Implantitis – Symptoms, Causes, Treatment Options & Cost, truy cập vào tháng 10 24, 2025, https://myimplantdentist.com.au/peri-implantitis/ 8. The Peri-Implantitis Guide: What It Is and Its Symptoms – Utah Periodontal Specialists, truy cập vào tháng 10 24, 2025, https://www.utahperio.com/blog/the-peri-implantitis-guide/ 9. Study Details | NCT06849856 | Evaluation of the Efficacy of Different Non-Surgical Peri-implantitis Treatment Approaches | ClinicalTrials.gov, truy cập vào tháng 10 24, 2025, https://clinicaltrials.gov/study/NCT06849856 10. Comparison of Air Abrasion and Mechanical Decontamination for Managing Inflammatory Reactions around Dental Implants: A Systematic Review and Meta-Analysis – MDPI, truy cập vào tháng 10 24, 2025, https://www.mdpi.com/2076-3417/14/17/7775 11. Predictive factors for the treatment success of peri-implantitis: a protocol for a prospective cohort study | BMJ Open, truy cập vào tháng 10 24, 2025, https://bmjopen.bmj.com/content/14/1/e072443 12. General recommendations in the non-surgical step of peri-implantitis treatment Recommendations for the surgical management of pe, truy cập vào tháng 10 24, 2025, https://www.efp.org/fileadmin/uploads/efp/Documents/Other_publications/Clinical_guidelines/peri-implantitis-guideline-03-peri-implant-diseases.pdf 13. Peri-implantitis Update: Risk Indicators, Diagnosis, and Treatment – PMC – NIH, truy cập vào tháng 10 24, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC7536094/ 14. Comparison of Non-Surgical Methods for Implant Surface Treatment in Simulated Bone Resorption Patterns: An In Vitro Study – MDPI, truy cập vào tháng 10 24, 2025, https://www.mdpi.com/2077-0383/14/20/7244 15. NON-SURGICAL TREATMENT OF PERI- IMPLANTITIS: CASE REPORT – Journal of IMAB, truy cập vào tháng 10 24, 2025, https://www.journal-imab-bg.org/issues-2023/issue2/2023vol29-issue2-4866-4868.pdf 16. Oral hygiene — key to dental implant longevity, truy cập vào tháng 10 24, 2025, https://implantpracticeus.com/ce-articles/oral-hygiene-and-dental-implant-maintenance-part-2/ 17. Detoxification of Implant Surfaces Affected by Peri-Implant Disease: An Overview of Non-surgical Methods – The Open Dentistry Journal, truy cập vào tháng 10 24, 2025, https://opendentistryjournal.com/VOLUME/8/PAGE/77/FULLTEXT/ 18. The Tipping Point for Air Polishing – Dimensions of Dental Hygiene, truy cập vào tháng 10 24, 2025, https://dimensionsofdentalhygiene.com/article/the-tipping-point-for-air-polishing/ 19. Air polishing revisited | Registered Dental Hygienists – RDH Magazine, truy cập vào tháng 10 24, 2025, https://www.rdhmag.com/pathology/periodontitis/article/16408826/air-polishing-revisited 20. (PDF) The use of non‐surgical interventions in patients with peri‐implantitis; a systematic review and meta‐analysis – ResearchGate, truy cập vào tháng 10 24, 2025, https://www.researchgate.net/publication/343640928_The_use_of_non-surgical_interventions_in_patients_with_peri-implantitis_a_systematic_review_and_meta-analysis 21. MECTRON Combi Touch – ariston dental, truy cập vào tháng 10 24, 2025, https://aristondental.com/mectron-combi-touch/ 22. Units – mectron dental, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/products/airpolishing/units/ 23. combi – mectron s.p.a. – PDF Catalogs | Technical Documentation, truy cập vào tháng 10 24, 2025, https://pdf.medicalexpo.com/pdf/mectron-spa/combi/73174-78825.html 24. combi touch – mectron dental, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/products/airpolishing/units/combi-touch/ 25. start your mectron, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/fileadmin/user_upload/dental/english/pdf/product_brochures/en_bro_start_your_mectron.pdf 26. Mectron-Combi-Touch-Brochure.pdf – ariston dental, truy cập vào tháng 10 24, 2025, https://aristondental.com/wp-content/uploads/2022/02/Mectron-Combi-Touch-Brochure.pdf 27. Good Practice in Non-surgical Periodontal Therapy – mectron dental, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/fileadmin/user_upload/dental/english/pdf/manuals/en_clin_book_nardi.pdf 28. start your mectron – Best Dental Shop, truy cập vào tháng 10 24, 2025, https://www.bestdentalshop.rs/download/combi-touch-katalog.pdf 29. Implant cleaning set S – mectron, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/l8/products/ultrasound/inserts-implant-cleaning/implant-cleaning-set-s/ 30. Piezosurgery Ultrasonic Implant Cleaning Kit (Mectron) | Dental Product, truy cập vào tháng 10 24, 2025, https://www.pearsondental.com/catalog/product.asp?majcatid=9465&catid=7859&subcatid=61579&pid=80137 31. Effectiveness of Erythritol-Based Air Polishing and Ultrasonic Instrumentation with PEEK Inserts in Peri-Implant Maintenance: A Randomized Clinical Trial Including Different Prosthetic Materials – MDPI, truy cập vào tháng 10 24, 2025, https://www.mdpi.com/2304-6767/13/6/235 32. PIEZOSURGERY® inserts for implant cleaning – MECTRON – Dentaltix, truy cập vào tháng 10 24, 2025, https://www.dentaltix.com/en/mectron/piezosurgeryr-inserts-implant-cleaning 33. Ultrasonic Implant Cleaning Tip F/ Mectron PiezoSurgery Woodpecker Ultrasurgery | eBay, truy cập vào tháng 10 24, 2025, https://www.ebay.com/itm/174920551997 34. Inserts implant cleaning – mectron dental, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/products/ultrasound/inserts-implant-cleaning/ 35. Prophylaxis powder sensitive+ – glycine powder – mectron, truy cập vào tháng 10 24, 2025, https://dental.mectron.com/products/airpolishing/powders/prophylaxis-powder-sensitive-glycine-powder/ 36. Air Polishing for Today's Dental Hygienist, truy cập vào tháng 10 24, 2025, https://dimensionsofdentalhygiene.com/article/air-polishing-for-todays-dental-hygienist/ 37. Clinical, microbiological and biochemical impact of a supportive …, truy cập vào tháng 10 24, 2025, https://pubmed.ncbi.nlm.nih.gov/36789504/ 38. Treatment of peri-implant mucositis using a glycine powder air-polishing or ultrasonic device. A randomized clinical trial | Request PDF – ResearchGate, truy cập vào tháng 10 24, 2025, https://www.researchgate.net/publication/274728709_Treatment_of_peri-implant_mucositis_using_a_glycine_powder_air-polishing_or_ultrasonic_device_A_randomized_clinical_trial 39. Evaluation of a single non-surgical approach in the management of peri-implantitis: glycine powder air-polishing versus ultrasonic device | Oral and Implantology, truy cập vào tháng 10 24, 2025, https://oimplantology.com/index.php/oimplantology/article/view/44 40. Differences Between Two Devices With Function of Periopolishing and Piezoelectric Scaler: A Randomized Clinical Trial – ResearchGate, truy cập vào tháng 10 24, 2025, https://www.researchgate.net/publication/389546916_Differences_Between_Two_Devices_With_Function_of_Periopolishing_and_Piezoelectric_Scaler_A_Randomized_Clinical_Trial 41. Differences Between Two Devices With Function of Periopolishing and Piezoelectric Scaler: A Randomized Clinical Trial – NIH, truy cập vào tháng 10 24, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11986920/ 42. Do air-polishing devices have any contraindications? | W&H, truy cập vào tháng 10 24, 2025, https://www.wh.com/en_global/dental-newsroom/reports-and-studies/new-article/1625470383252 43. Scholars Journal of Dental Sciences (SJDS) Air Polishing–An Overview – SAS Publishers, truy cập vào tháng 10 24, 2025, https://saspublishers.com/media/articles/SJDS_53_139-142_c.pdf